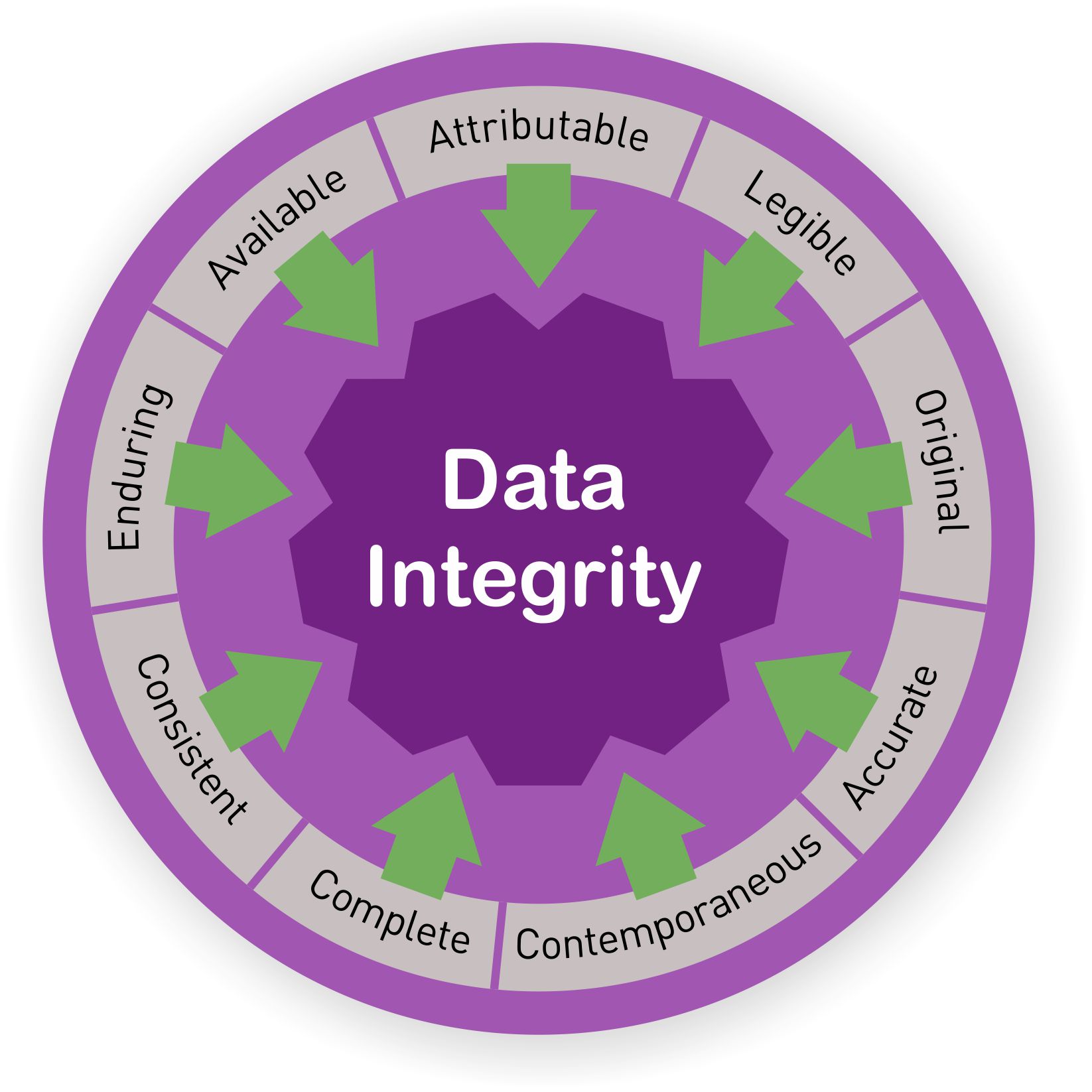
What Are The Principles That Ensure Data Integrity? Overall, data integrity is key to regulatory compliance, quality assurance, and the company's reputation within the industry. 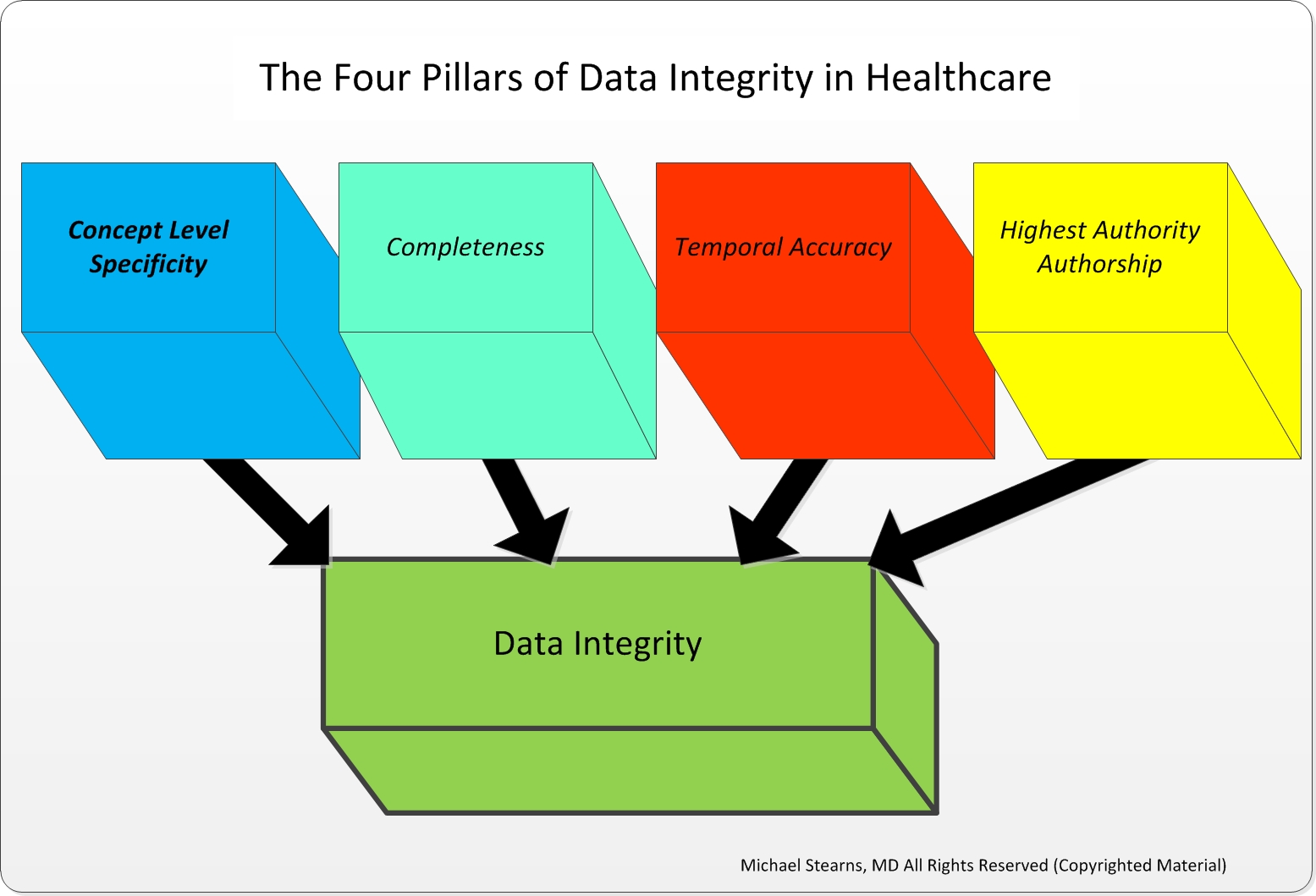
With data integrity in place, there is no need to inspect every process involved in drug production and supply, thus saving time and money.Īdditionally, data integrity guarantees trust between regulatory agencies and the industry as a whole. The safety, efficacy, and quality of products can be ensured with accurate, reliable, and valid data provided by data integrity compliance. Today, data is the backbone of compliance, efficiency of innovations, and quality assurance. Why is Data Integrity Important?įor the past several years, FDA and other global regulatory bodies have been increasing attention to data integrity problems in various industries. Overall, data quality is an essential part of data integrity. To be considered quality, data must be complete, valid, and consistent.ĭata integrity requires data quality to be in context to ensure usability to the organization. Data Integrity vs Data Qualityĭata quality is the reliability of data. It's the assurance that data is unchanged from its source and hasn't been modified in an unauthorized way. Data validation is the process of checking data quality and accuracy before using.ĭata Integrity - Data integrity is the completeness, soundness, and wholeness of data that complies with the intention of its creators. While data integrity and data validity may appear similar, they are not interchangeable terms.ĭata Validity - Data validity is correctness and reasonableness. 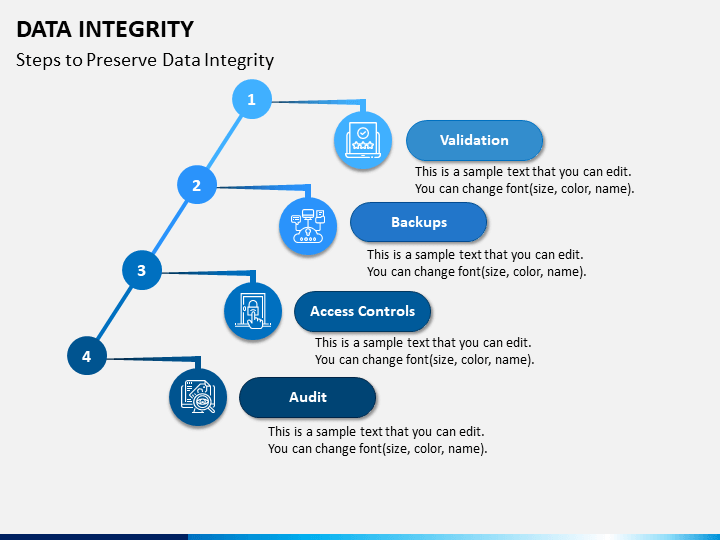
It can cause harm to the end user, result in incorrect decisions, initiate product recalls, and affect the company's reputation. Poor data integrity doesn't just result in regulatory non-compliance issues and penalties. Data integrity is critical in FDA-regulated industries since it makes sure that products meet the required quality standards. What Is Data Integrity?ĭata integrity is accuracy, reliability, and consistency of data throughout its lifecycle. This article will talk define data integrity and explain how to maintain it effectively. Meanwhile, lack of data integrity could result in Form 483 observations, a Warning Letter, and potential recall of products that fail to meet quality standards or requirements. To ensure compliance, FDA regulates data integrity in several ways through the GMP, GCP, GLP (GxP) regulations and 21 CFR Part 11 requirements.Īll the data-related FDA requirements can be addressed by maintaining good data integrity practices. Integrity, security, and consistency of data lie at the core of FDA-regulated company operations. Besides being a requirement for compliance reasons, data integrity is the foundation of a top-notch quality management system and paramount for ensuring patient safety. Food and Drug Administration)-regulated industries, organizations, and projects, the importance of data integrity is extremely high.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
